 Lanthanides: The third group is much too long to fit into the third column, so it is broken out and flipped sideways to become the top row of the island that floats at the bottom of the table. Their chemical reactions typically occur more slowly and produce less heat compared to the alkali metals. But they're not as reactive as the alkali metals. Each of these elements has two electrons in its outermost energy level, which makes the alkaline earths reactive enough that they're rarely found alone in nature. Hydrogen, with its single electron, also lives in Group 1, but the gas is considered a nonmetal.Īlkaline-earth metals: The alkaline-earth metals make up Group 2 of the periodic table, from beryllium (Be) through radium (Ra). They are also extremely reactive and will burst into flame or even explode on contact with water, so chemists store them in oils or inert gases. Shiny and soft enough to cut with a knife, these metals start with lithium (Li) and end with francium (Fr). Available here Image Courtesy:ġ.The periodic table of elements is arranged into several broad groups (Image credit: Future) Groups of the Periodic tableĪlkali metals: The alkali metals make up most of Group 1, the table's first column. “Chemistry for Non-Majors.” Lumen, Lumen Learning. “The Difference Between an Element Group and Period.” ThoughtCo, Aug. And, the key difference between periods and groups is that periods are horizontal rows whereas groups are the vertical columns in the periodic table of chemical elements. To sum up what we discussed here, periods and groups are two ways that we categorize the chemical elements in the periodic table. That is, the elements in the same period have the same number of electron shells while the elements in the same group have the same number of valence electrons. Furthermore, we can also find another difference between periods and groups in the eletron arrangement. There are 7 major periods and 18 groups in the periodic table of elements. Hence, this is the key difference between periods and groups of the periodic table. Periods are the horizontal rows in the periodic table whereas groups are the vertical columns in the periodic table. What is the Difference Between Periods and Groups in Periodic Table? For example, we call the group 1 element as alkali metals and group 2 elements as alkali earth metals. However, some groups have common names as well. There are mainly 18 groups in the periodic table. 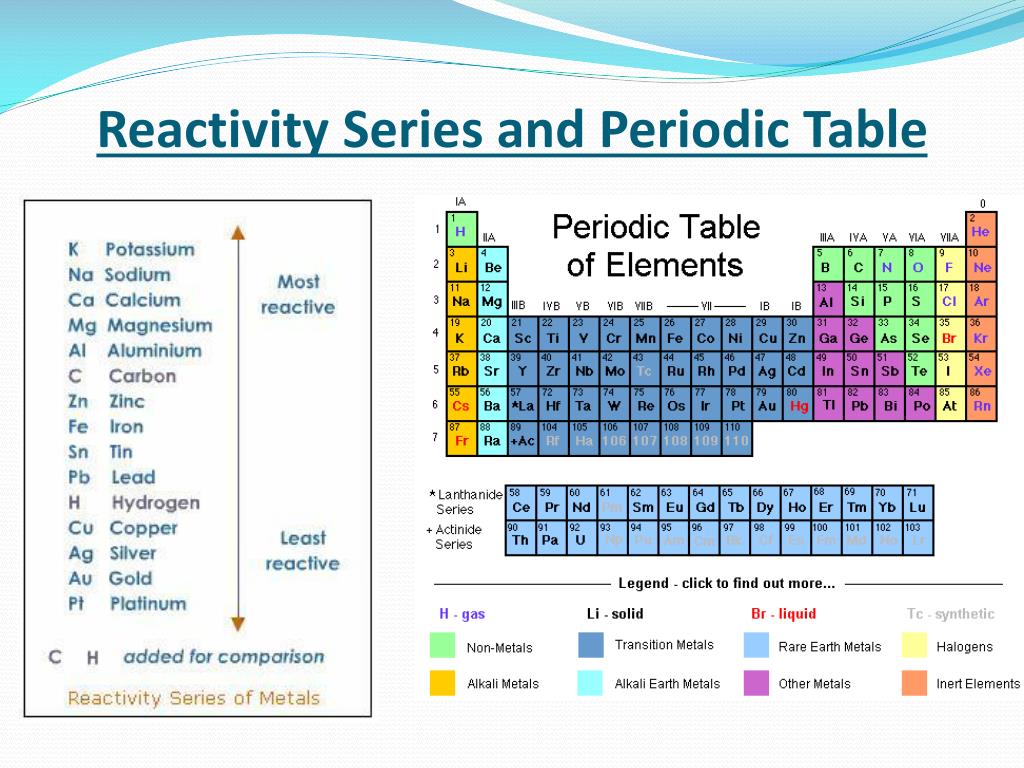 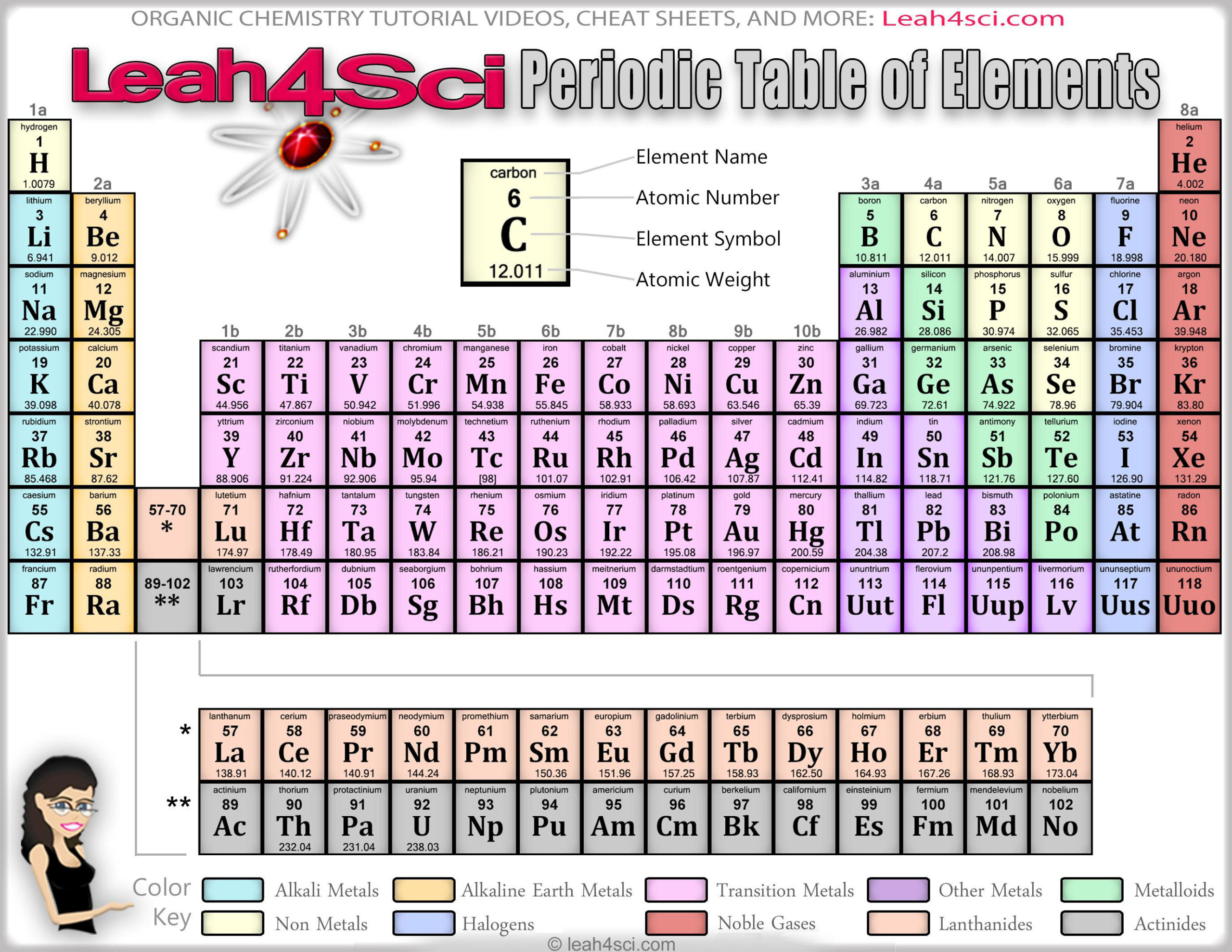
Most of the times, the elements in the same group shares somewhat similar chemical and physical properties. For example, the number of valence electrons in the group 1 is 1. All the chemical elements in the same group contain an equal number of valence electrons. Groups are the vertical columns in the periodic table. Thus, we name them as period 1, period 2, … Period 7. Mainly, there are 7 periods in the periodic table. This is because, the number of elements is determined by the number of electrons allowed in each electron shell. However, the periods have different numbers of members there are more chemical elements in some periods than some other periods. Moreover, the elements get less metallic when we move forward a row. When we go through a row, the number of protons in the atomic nucleus increases by 1 for each next element in the period. All the chemical elements in this row have the same number of electron shells. Periods are the horizontal rows in the periodic table. Summary What are Periods in Periodic Table? Side by Side Comparison – Periods vs Groups in Tabular Formĥ. Further, each row and column has specific characteristics, and we name a row as a period and a column as a group in the periodic table. Hence, as with any grid, this periodic table also has rows and columns. 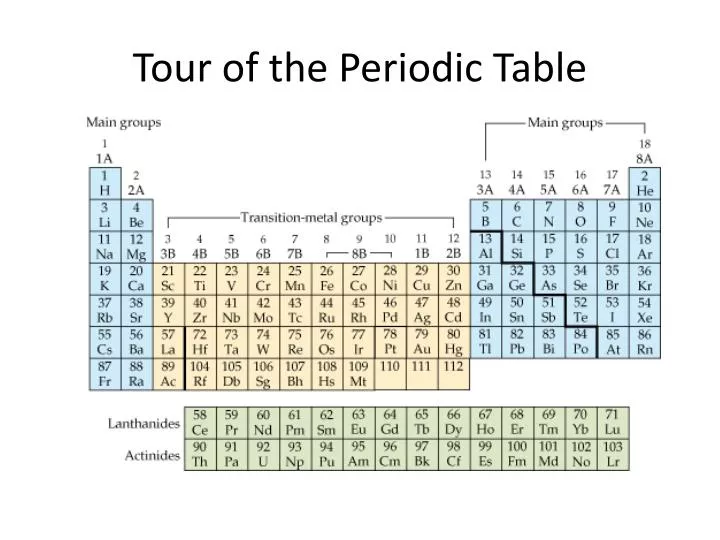
The periodic table of elements is a large table in which each and every known chemical element is placed in a specific location considering the atomic structures. The key difference between periods and groups is that the periods are horizontal rows whereas the groups are the vertical columns in the periodic table of chemical elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
